- Home
- / Insights
- / Whitepapers
- / Looking Ahead Risk Outlook Medical Supply Chain
Looking Ahead: Risk Outlook for the Medical Device Supply Chain
-
January 12, 2023
DownloadsDownload White Paper
-
Delayed and cancelled elective surgeries, long lead times, significantly higher costs – this has been the unfortunate state of affairs. COVID-19 spared no industry, and the medical device industry was especially impacted, even while healthcare needs around the world increased. Extensive and unprecedented supply chain disruptions have materialized and sustained over the last 30 months, including raw material shortages, labor challenges, sterilization constraints, and concerns surrounding device security and cybersecurity.
Healthcare and Life Sciences Quarter in Review Read Now
Industry leaders have been pressed to adapt to the challenges by reshoring manufacturing capacity, building contingency in multiple levels of the supply chain, and driving sustainable improvement to overcome long-lasting issues around labor, cost increases, raw material supply disruptions and increased demand.
FTI Consulting explores the challenges that the industry, still struggling to regain stability, confronts over the upcoming months and years. Many of the challenges represent significant and potentially enduring changes to policy, resource availability and accepted practice.
Labor
Remote Work
There is overwhelming evidence that much of the workforce in the United States enjoys and prefers working from home. The U.S. Bureau of Labor Statistics indicated that even before the COVID-19 pandemic, nearly 16% of the US workforce worked remotely; at the height of the pandemic, that number had significantly increased to a remarkable 71%. According to a 2022 Pew Research Center Study, of Americans with ability to work from home, 59% choose to do so nearly all the
time.1,2
While the desire to work remotely is accelerating, a majority of those employed in the healthcare industry do not have that luxury, and those in manufacturing have even fewer opportunities for remote work.
Medical device manufacturers have been forced to make significant changes to accommodate a remote workforce where possible:
- Implemented digital infrastructure to allow those not on the shop floor to work remotely.
- Improved clarity and measurement of goals and objectives to communicate expectations and increase employee accountability.
- Emphasized “relational management” to compensate for the lack of social interaction.
- Explored ways to utilize best-cost countries such as Columbia, Poland, and India for remote work.
Despite these alterations, Medtech companies continue to struggle with staffing technical positions.3
A Dearth of Skilled Technicians
The outlook for the industry is positive. However, the current and projected availability of skilled technicians does not match the projected demand.
There is a bolus of programmers and machinists at retirement age. On top of that, the push for college degrees and the societal downplaying of skilled labor over the course of the past 20 years has reduced the numbers of young workers entering this space according to Shannon Mills, Senior Director in FTI Consulting’s Supply Chain practice.
The lack of skilled tehnicians is most severley felt outside of the medical device clusters. Massachusetts, for example, attracts and retains medical device manufacturers, in part due to the high wages and the potential for career mobility.3 Desmond Adler, President of Cardiovascular at Gentuity, states that despite labor costs being so high in the New England area, he has been able to find skilled technicians to manufacture their complex catheters. While he is worried about the next ten years, he has no immediate concerns surrounding labor costs or availability. While this may be a relief for nimble, smaller-sized firms, industry giants such as Medtronic and Zimmer Biomet still have many open positions.
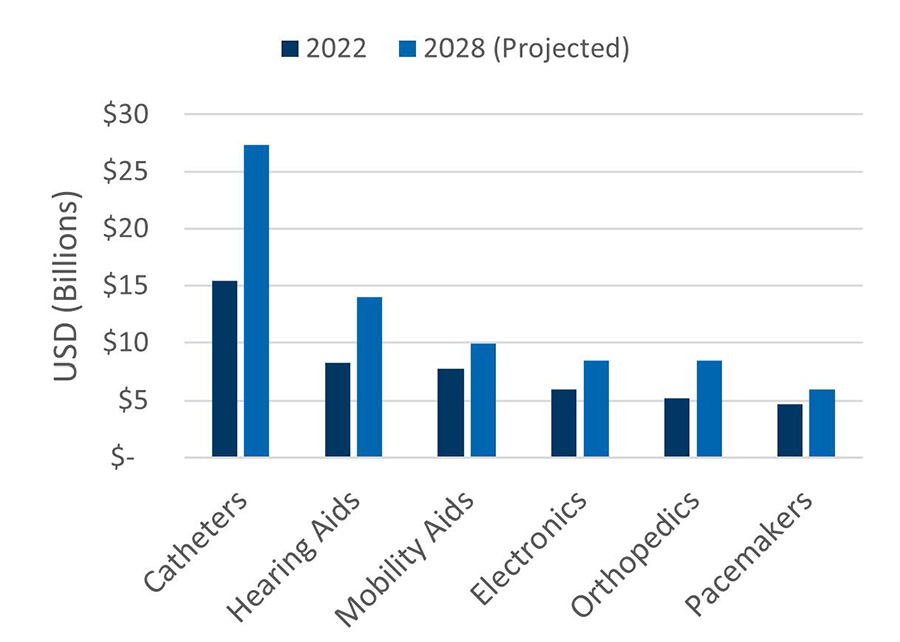
Global Medical Device Market
Medical Device
Raw Materials
Alloys and metals, such as titanium, cobalt-chrome and plastics, are commonly used in producing millions of medical devices and implants for joint replacement surgeries. Geopolitical volatility, energy crises, health concerns, and several other factors inhibit continuous supply of raw materials to the medical device industry. As the demand for medical devices continues to increase, manufacturers face major disruptions and long lead times.
Disruption of Alloy Imports
Disruptions to the global markets amidst the Russian-Ukrainian War have caused uncertainties surrounding the supply of critical raw materials utilized in medical device production. These uncertainties stem from import restrictions, concentration of production in only a few countries, and decades of bilateral dependency.4
The Chip Shortage
Pandemic-driven lockdowns in early 2020 drove down economic activity. Consumers avoided the purchase of cars, computers, and other technologies. Elective surgeries were pushed out or cancelled altogether. Accordingly, production decreased, and chip manufacturing, particularly in Asia, grinded to a near complete halt.
Fast forward to the end of 2020, consumers started buying cars and consumer electronics, medical device surgeries picked up, and the raw material purchases for production became extremely fast-tracked. Meanwhile, Asia’s continued battle with COVID-19 and its variants added pressure to an already constrained supply of chips.5
An April 2022 study conducted by Deloitte presented a harsh reality for medical device manufacturers: 6
- More than half of respondents single-sourced their semiconductors at the time of the study, and all were pursuing alternative sources.
- A third of respondents resorted to brokers for semiconductor supply as opposed to directly engaging suppliers.
- Prior to the shortage, a majority of the medical device manufacturers did not have a large chip inventory on hand; subsequently over 70% committed to increasing inventory levels.
Looking forward to next year, the global semiconductor chip industry is expected to surpass $600 billion USD but a single chip, worth just a few dollars, may inhibit the sale of pricey (and critical) medical devices.
Rick van der Vegte, Managing Director in FTI’s Supply Chain practice, suggests that small companies consider buying cooperatives: “Shortages cause huge fluctuations in demand and supply, because it stimulates ‘hoarding’ behavior. Suddenly the large orders get delivered to those who had the capital to place them in the first place… The ripple effect is big.”
Sterilization Constraints
The medical device industry heavily relies on sterilization to bring its products to market. Companies rely on ethylene oxide (EtO), a leading method of sterilization due to its inert chemical properties, thereby preventing polymer degradation. EtO is used to sterilize over 50% of products (over 20 billion units) in the United States.7
There is significant pressure to reduce EtO sterilization since it is a human carcinogen and has been linked to adverse health effects. Recent lawsuits against sterilization companies using EtO in the United States have challenged the sterilization industry, as will impending updates to the Clean Air Act’s National Emission Standards for Hazardous Air Pollutants (NESHAP).8
Recently, in September 2022, an Illinois jury ruled against major sterilization company Sterigenics, awarding a $363 million verdict for EtO emissions tied to a plaintiff’s cancer diagnosis. This was just one of many personal injury lawsuits filed against Sterigenics’ parent company, Sotera Health. Needham & Company analysts warn that additional lawsuits may be filed against sterilization facilities, as well as major medical device companies.9
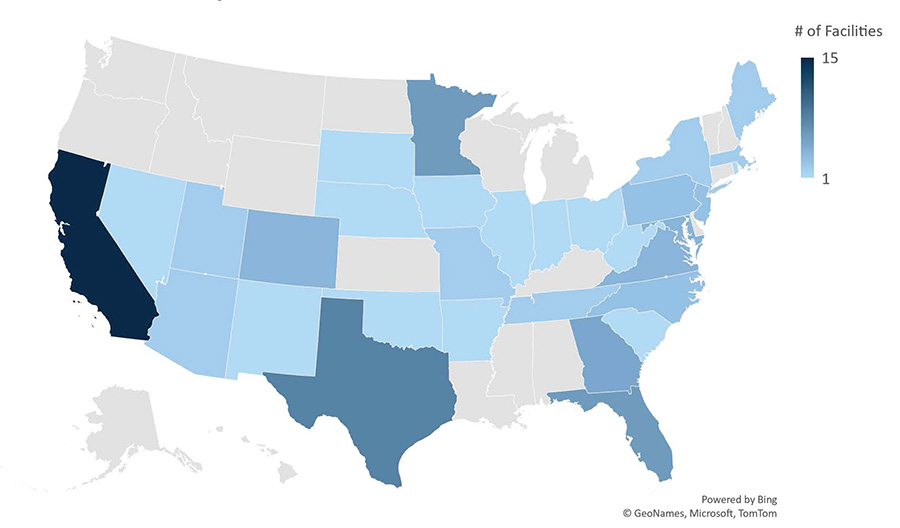
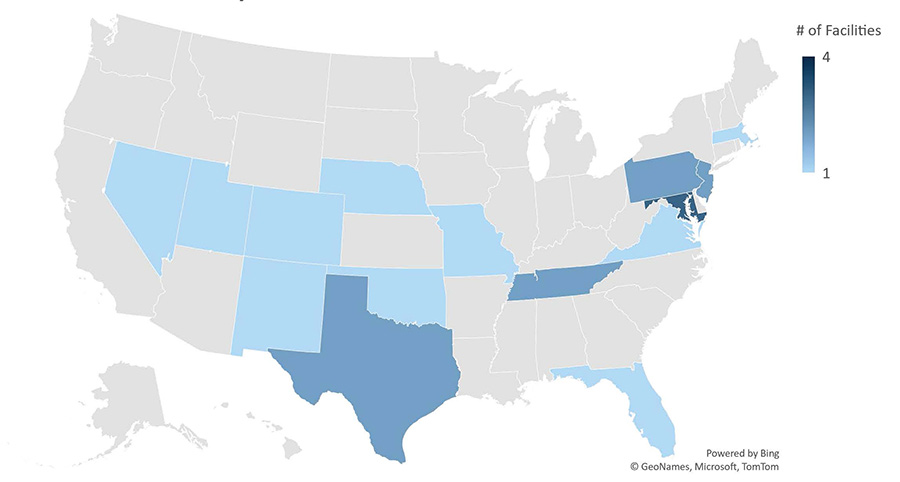
As of October 4, 2022, 96 commercial sterilization facilities in the United States utilize EtO.10 The EPA found that 25 of these facilities emit excessive levels of EtO (at or above the level of 100 additional cancer cases per million people exposed). EPA is in the process of updating the National Emission Standards for Hazardous Air Pollutants (NESHAP) under the Clean Air Act, in order to reduce the risk from EtO.11
Ethylene Oxide Commercial Sterilization Facilities
Data as of 10/04/2022, Source: U.S. Environmental Protection Agency12
Ethylene Oxide Commercial Sterilization Facilities Evaluated by FDA and Found with Excessive Emissions
Data as of 10/04/2022, Source: U.S. Environmental Protection Agency13
Companies will need to consider reducing EtO emissions and explore alternative sterilization methods in response to legal and environmental groups. While this may pose a challenge, companies might save resources in the long run.
The FDA is putting additional pressure on sterilization capacity with increased scrutiny relating to high-level disinfection. The agency is discouraging use of high-level disinfection (HLD) for certain medical devices, pushing providers to use more effective sterilization methodologies. Smaller providers that do not have their own sterilization equipment must compete with already limited availability.
There are other alternative sterilization methods, which include heat (in the form of steam), dry heat, radiation, vaporized hydrogen peroxide, and other gases. Genuity’s Desmond Alder shared that EtO is not used in sterilizing their catheter. He exclaimed that challenging or pushing back against the EPA is not worth the time and effort and instead utilizes other methods of sterilization.
David Linehan, Vice President of Business Development at Bio-One Sciences states that his company’s technology uses ultraviolet-C (UVC) light for high level disinfection in clean rooms. The robot uses a controlled dose of UV light to sterilize against viruses and bacteria. The technology has not yet been used broadly to reprocess or sterilize medical devices directly.
Cybersecurity Concerns
Cybersecurity, a serious concern in any industry, is particularly acute in healthcare. The sensitivity of Personal Health Information (PHI) invokes stringent privacy laws, and the Internet of Things (IoT) opens patients to risk of direct intervention with a medical device’s provision of life-saving therapies. The potential risks to patient safety grow as the healthcare industry increases their reliance on digital technology for diagnostics, record storage, treatment, and practice management. Just this year, Quincy, Massachusetts’ Shields Healthcare Group, detected a breach from malware that involved PHI of over 2 million of their patients.14
A cybersecurity investigation conducted by FTI Consulting highlighted that, in the last few years, the magnitude of exposed records and financial losses due to data breaches have increased rapidly.15 Despite this being a rising issue in the industry, the cybersecurity strategies of most hospitals are still focused on traditional, centralized information security, which leaves most medical devices vulnerable to attack.16 As these devices become more advanced in their capability and diverse in their application, there is an increased need for enhanced security when it comes to patients’ information and treatment. Consequences of cyberattacks in the past year have included poor patient outcomes due to procedure or test delays, longer hospital stays, and heightened mortality rates, which makes cybersecurity an important priority to defend against them.17
The FDA has declared that they will only clear, authorize, and approve medical devices for use if they deem that there is reasonable assurance that they are safe and effective at a broad level.18 However, these devices have a multi-layered supply chain of vendors, as they are often comprised of multiple hardware, firmware, and software components.19 In recent years, supply chain disruptions have driven manufacturers to pursue new suppliers. Changing vendors opens manufacturers up to more vulnerabilities because each new supplier needs to be vetted to build trust regarding the quality and security of their products. This makes it difficult for manufacturers to ensure that uniform and sufficient cybersecurity is integrated into every step in the lifecycle of a device.
Medical device manufacturers (MDMs) are responsible for identifying cybersecurity risks and vulnerabilities associated with their individual devices.20 The FDA is responsible for providing guidelines and regulations for the security of these devices, but these are often the minimum threshold needed for safety, meaning that many entities that are compliant are not necessarily secure.21 The FDA actively communicates with members in the industry to set up regulatory action, but their efforts often have difficulty keeping up with technological innovation and advancement in the industry. In 2022, it was found that around 70% of medical devices like MRI machines, heart rate monitors, medication pumps, etc. are running outdated, unpatched operating systems with critical vulnerabilities which are welcoming entryways for attackers.22
As early as 2013, Massachusetts had seen that unpatched devices are problematic when Boston’s Beth Israel Deaconess Medical Center found that 664 of their medical devices were running outdated operating systems and had to take a couple devices offline each week to remove malware.23 Even since then, a 2020 study by Ponemon Institute found that half of hospitals have been attacked with ransomware, and that 88% of the attacks involved IoMT (internet of medical things) devices like with immature and unpatched cybersecurity infrastructure.24
While the health care environment as an entity is complex, manufacturers, providers, and regulatory agencies alike have to work together to manage cybersecurity risks.25 The FDA has begun to make strides in this direction by asking Congress for approximately $5.5 million to develop and staff their own cybersecurity program for devices in 2023 and having them codify their authority to implement cybersecurity requirements into their approval process through The PATCH Act.26,27 With this funding and authority, the FDA will be able to have more personnel focus on ensuring sufficient cybersecurity in medical devices throughout their life cycles, hopefully mitigating the number of medical devices with product security concerns that not only make it to market but remain on the market.28
Conclusion
Presently, the medical device industry is facing a unique and evolving set of supply chain challenges across the United States and around the world. Whether pandemic-related or the byproduct of technological advancement, these phenomena require close monitoring and decisive action in order to meet market demand.
Our Expert Insights:
- Continue to build company culture to motivate workers, increase transparency with evaluation measures to maintain employee accountability, and retain skilled technicians. Approach technology as a tool to enhance job performance and look into trainings intended at building knowledge.
- Stay vigilant of market surpluses and shortages (alloys, chips, etc.) and assess opportunities for cooperative buying to ensure more steady supply, reduce sourcing reexaminations, and stabilize supply and demand fluctuations overall.
- Keep abreast of EtO emissions levels and ensure they are in line with EPA standards, reevaluating sterilization methods and considering alternatives to eliminate risk of adverse health effects and prevent legal complications in the future.
- Prioritize patient data security during the development and testing process for increasingly advanced medical devices with network connectivity capabilities. Revisit cybersecurity protocols already in place and bolster risk identification systems to proactively reduce incidents.
Each of the four topics discussed traces back to the importance of adaptability in an ever-changing world. As market trends, regulations, and environmental factors evolve, so must organizations and their supply chains – or risk getting mired in the ways of the past.
Footnotes:
1: Kim Parker, Juliana Menasce Horowitz, and Rachel Minkin, “COVID-19 Pandemic Continues To Reshape Work in America,” Pew Research Center (February 16, 2022), https://www.pewresearch.org/social-trends/2022/02/16/covid-19-pandemic-continues-to-reshape-work-in-america.
2: Dalton, Michael. “Telework during the COVID-19 Pandemic: Estimates Using the 2021 Business Response Survey : Monthly Labor Review.” U.S. Bureau of Labor Statistics. U.S. Bureau of Labor Statistics, March 2022. https://www.bls.gov/opub/mlr/2022/article/telework-during-the-covid-19-pandemic.htm#:~:text=At%20the%20time%20of%20the,involving%20telewo rking%20rarely%20or%20never
3: Stanton, Kelly. “Industry Voices-in a Troubled Economy, Emerging Medical Device Companies Need to Double Down on Driving Efficiencies.” Fierce Healthcare, September 23, 2022. https://www.fiercehealthcare.com/health-tech/industry-voices-troubled-economy-emerging-medical-device-companies-need-double-down.
4: Moise, Evdokia. “The Supply of Critical Raw Materials Endangered by Russia's War on Ukraine.” OECD. Accessed November 3, 2022. https://www.oecd.org/ukraine-hub/policy-responses/the-supply-of-critical-raw-materials-endangered-by-russia-s-war-on-ukraine-e01ac7be/
5: J.P. Morgan Chase. “How Long Will the Chip Shortage Last?: J.P. Morgan Research.” J.P. Morgan. J.P. Morgan Chase, August 11, 2022. https://www.jpmorgan.com/insights/research/supply-chain-chip-shortage
6: Stewart, Duncan, Dan Hamling, Ariane Bucaille, and Gillian Crossan. “My Kingdom for a Chip: The Semiconductor Shortage Extends into 2022.” Deloitte Insights. Deloitte, November 30, 2021. https://www2.deloitte.com/xe/en/insights/industry/technology/technology-media-and-telecom-predictions/2022/semiconductor-chip-shortage.html
7: “Ethylene Oxide Risk from Commercial Sterilizers.” EPA. Environmental Protection Agency, October 16, 2022. https://www.epa.gov/hazardous-air-pollutants-ethylene-oxide/forms/ethylene-oxide-risk-commercial-sterilizers
8: Jim Hammerand, “EPA flags high-cancer-risk EtO sterilization facilities across the country,” Medical Design & Outsourcing (August 3, 2022), https://www.medicaldesignandoutsourcing.com/epahigh-cancer-risk-ethylene-oxideeto-sterilization-facilities-us/.
9: Ricky Zipp, “Device makers with ethylene oxide facilities at risk of lawsuits after Sterigenics loss: Needham,” MedTech Dive (September 23, 2022), https://www.medtechdive.com/news/device-companies-lawsuits-ethylene-oxide-sterigenics/632545/.
10: “Ethylene Oxide Commercial Sterilization Facilities,” United States Environmental Protection Agency (last updated on August 17, 2022), https://www.epa.gov/hazardous-air-pollutants-ethylene-oxide/ethylene-oxide-commercial-sterilization-facilities.
11: “Ethylene Oxide Risk from Commercial Sterilizers,” United States Environmental Protection Agency (last updated on August 16, 2022), https://www.epa.gov/hazardous-air-pollutants-ethylene-oxide/forms/ethylene-oxide-risk-commercial-sterilizers.
12: Ibid.
13: Ibid.
14: Marianne Kolbasuk McGee, “Hack of Medical Imaging Provider Affects Data of 2 Million,” Healthcare Info Security (June 7, 2022), https://www.healthcareinfosecurity.com/hack-medical-imaging-provider-affects-data-2-million-a-19227.
15: Matt Chevraux, Juliana Malhotra, and Rajdeep Mukherjee, “Safeguarding Protected Health Information (PHI): Preparing for the Onslaught,” FTI Consulting, Inc. (October 19, 2022), https://www.fticonsulting.com/insights/articles/safeguarding-protected-health-information-phi.
16: Doug Folzom, “Medical Device Cybersecurity Hinges on Far More Than Identifying Risks,” HIMSS (last visited October 28, 2022), https://www.himss.org/resources/medical-device-cybersecurity-hinges-far-more-identifying-risks.
17: “Cyber Insecurity in Healthcare: The Cost and Impact on Patient Safety and Care,” Proofpoint (last visited November 02, 2022), https://www.proofpoint.com/us/cyber-insecurity-in-healthcare.
18: Ibid.
19: Sean Ashcroft, “Medical devices supply chains ‘facing cybersecurity storm,’” SupplyChain (February 17, 2022), https://supplychaindigital.com/supply-chain-risk-management/medical-devices-supply-chains-facing-cybersecurity-storm.
20: “Cybersecurity,” U.S. Food & Drug Administration (content current as of October 7, 2022), https://www.fda.gov/medical-devices/digital-health-center-excellence/cybersecurity.
21: Mike Kijewski, “Medical Device Manufacturers Need To Act As Regulators Sharpen Their Cybersecurity Guidelines,” Forbes (June 10, 2022), https://www.forbes.com/sites/forbestechcouncil/2022/06/10/medical-device-manufacturers-need-to-act-as-regulators-sharpen-their-cybersecurity-guidelines/?sh=1e411915dd63.
22 Eric Henry, “Designing Secure Medical Devices: Building Cybersecurity into the Development Process,” FDANews (January 2022), https://www.fdanews.com/products/62026.
23: Katherine Booth Wellington, “Cyberattacks on Medical Devices and Hospital Networks: Legal Gaps and Regulatory Solutions,” Santa Clara High Technology Law Journal, Article 1, Volume 30, Issue 2 (January 2013), https://digitalcommons.law.scu.edu/cgi/viewcontent.cgi?referer=&httpsredi r=1&article=1578&context=chtlj.
24: Milliard, Mike. “Direct Line between Hospital Cyberattacks and Patient Mortality, Report Shows.” Healthcare IT News, September 12, 2022. https://www.healthcareitnews.com/news/direct-line-between-hospital-cyberattacks-and-patient-mortality-report-shows
25: McKeon, Jill. “FDA, OIG Request Cybersecurity Investments in FY 2023 Budget.” HealthITSecurity. HealthITSecurity, March 30, 2022. https://healthitsecurity.com/news/fda-oig-request-cybersecurity-investments-in-fy-2023-budget.
26: Ibid.
27: Ibid.
28: https://www.healthcareitnews.com/news/patch-act-seeks-shore-security-medical-devices-iot-networks
Related Insights
Related Information
Published
January 12, 2023
 Key Contacts
Key Contacts
Senior Consultant